Leading global biopharma outsourcing company adopts Veeva Vault Study
Startup following its success with Veeva Vault eTMF
PHILADELPHIA--(BUSINESS WIRE)--
2016 Veeva R&D Summit – Today at the Veeva
R&D Summit, to an audience of more than 600 industry leaders, Veeva
Systems (NYSE:VEEV) today announced that inVentiv Health, a leading
biopharma outsourcing company, is expanding its adoption of the Veeva
Vault Clinical Suite to expedite site activation for clients.
inVentiv Health – comprised of a top-tier full-service CRO and the
industry’s only Contract Commercial Organization (CCO) – is building on
its successful use of Veeva
Vault eTMF by adopting Veeva
Vault Study Startup to accelerate document-intensive site activation
and speed time to first patient enrollment.
This Smart News Release features multimedia. View the full release here:
http://www.businesswire.com/news/home/20161018005685/en/
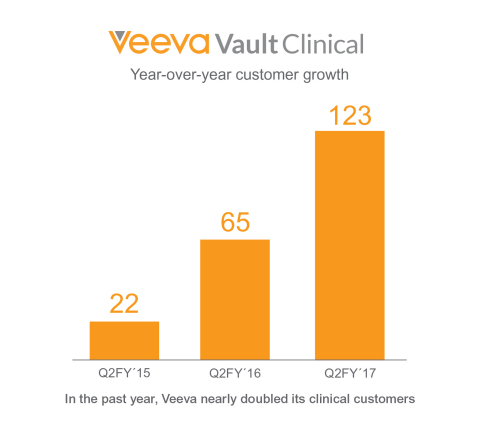
(Graphic: Business Wire)
“We recognized the value an innovative technology solution could have in
knocking down hurdles that slow progress during the study start-up
phase, where improvements can have a significant impact on overall trial
results,” said Rachel Stahler, CIO, inVentiv Health. “inVentiv
identified Veeva as a best-of-breed platform that can maximize
efficiencies and accelerate the delivery of much needed drugs to
patients.”
Vault Study Startup will help inVentiv streamline complex activities
related to site activation, including ethics committee and Ministry of
Health submissions and approvals, document distribution and tracking,
and site contract transactional activities. Now inVentiv can improve the
efficiency of trial processes by managing documents, data, and study
start-up activities on a single cloud platform, as well as provide
stakeholders easy access to the same, up-to-date clinical information
for improved collaboration. In addition, seamless interoperability
between Vault Study Startup and Vault eTMF ensures that documents and
data are available in both applications.
The current clinical operations environment is highly complex and often
includes upwards of 20 disparate systems to manage trials – from
electronic data capture (EDC) systems and study start-up applications to
clinical trial management systems (CTMS) and electronic trial master
files (eTMF). Legacy technologies limit efficiency, visibility, and
collaboration between sponsors, investigator sites, and CROs. In fact,
in a recent industry-wide survey, nearly half (49%) of all respondents
cited integration between their eTMF and CTMS applications as a key,
unmet need.1
Kristen Jackson, senior director, Essential Document Collection and
Submissions, Site Centricity Unit, inVentiv, will be a speaker at the
2016 Veeva Global R&D Design Summit in Philadelphia on October 18 and
will discuss these challenges and the benefits of Vault Study Startup.
About Veeva Vault Clinical Suite
The Veeva Vault Clinical Suite is the only unified suite of applications
on a single cloud platform that streamlines clinical operations and data
management, from study start-up to archive. Veeva’s clinical operations
solutions include Vault eTMF, Vault Study Startup, and Vault
CTMS, while newly announced Veeva
Vault EDC and Veeva
Vault eSource will support clinical data management. Read our press
release to learn more about Vault EDC and Vault eSource and how
Veeva will transform clinical data management by delivering real-time,
accurate data to help enable faster, more informed decisions in clinical
trials.
Additional Information
For more on Veeva Vault Study Startup, visit: veeva.com/studystartup
For more on Veeva Vault eTMF, visit: veeva.com/eTMF
Connect with Veeva on LinkedIn: linkedin.com/company/veeva-systems
Follow us @veevasystems on Twitter: twitter.com/veevasystems
Like Veeva on Facebook: facebook.com/veevasystems
About Veeva Systems
Veeva Systems Inc. is a leader in cloud-based software for the global
life sciences industry. Committed to innovation, product excellence, and
customer success, Veeva has more than 450 customers, ranging from the
world's largest pharmaceutical companies to emerging biotechs. Veeva is
headquartered in the San Francisco Bay Area, with offices in Europe,
Asia, and Latin America. For more information, visit www.veeva.com.
Forward-looking Statements
This release contains forward-looking statements, including the market
demand for and acceptance of Veeva’s products and services, the results
from use of Veeva’s products and services, and general business
conditions, particularly in the life sciences industry. Any
forward-looking statements contained in this press release are based
upon Veeva’s historical performance and its current plans, estimates,
and expectations, and are not a representation that such plans,
estimates, or expectations will be achieved. These forward-looking
statements represent Veeva’s expectations as of the date of this press
announcement. Subsequent events may cause these expectations to change,
and Veeva disclaims any obligation to update the forward-looking
statements in the future. These forward-looking statements are subject
to known and unknown risks and uncertainties that may cause actual
results to differ materially. Additional risks and uncertainties that
could affect Veeva’s financial results are included under the captions,
“Risk Factors” and “Management’s Discussion and Analysis of Financial
Condition and Results of Operations,” in the company’s filing on Form
10-Q for the period ended July 31, 2016. This is available on the
company’s website at veeva.com
under the Investors section and on the SEC’s website at sec.gov.
Further information on potential risks that could affect actual results
will be included in other filings Veeva makes with the SEC from time to
time.
1 Veeva 2016 Paperless TMF Survey: Annual Report (Veeva: June
2016)

View source version on businesswire.com: http://www.businesswire.com/news/home/20161018005685/en/
Source: Veeva Systems Inc.