Veeva Vault Submissions and Veeva Vault QualityDocs help ICON
increase visibility and improve collaboration with sponsors
PHILADELPHIA--(BUSINESS WIRE)--
2016 Veeva R&D Summit - Today at the Veeva
R&D Summit, to an audience of more than 600 industry leaders, Veeva
Systems (NYSE:VEEV) today announced that ICON, a global provider of
drug development solutions and services, is streamlining its regulatory
and quality operations with Veeva Vault applications to increase
transparency and enable greater collaboration with sponsors.
This Smart News Release features multimedia. View the full release here:
http://www.businesswire.com/news/home/20161018005699/en/
(Graphic: Business Wire)
Veeva Vault provides ICON an integrated suite of best-in-class
applications that unify content and data for improved quality and
compliance across their global operations. Veeva
Vault Submissions delivers a single, authoritative source for ICON
and their affiliates and partners to speed the entire regulatory
submission preparation process – from authoring to assembly. Separately, Veeva
Vault QualityDocs improves GxP document management for greater
compliance, improved quality, and reduced operational overhead.
Communication, visibility, and process inefficiency are common
challenges between CROs and sponsors when data and content are managed
in multiple, disconnected systems. This hinders collaboration and often
creates duplicate work and content among teams, introducing compliance
risks that can delay time to market.
With Veeva Vault at the heart of its regulatory and quality operations,
ICON has a single source of truth for regulatory information and key
quality policies and procedures that global stakeholders can share in
real time. Now ICON can provide its sponsors and partners with full
visibility into regulatory and quality content for increased control and
collaboration.
“Veeva Vault applications enable ICON to more easily collaborate with
sponsors on all documents throughout product development lifecycles and
streamline communications to shorten delivery timeframes,” said Quintin
van Wyk, vice president, safety, regulatory, and writing services for
ICON.
Faster, more efficient global submissions process
Vault Submissions will be part of ICON’s medical writing and regulatory
services as well as submissions management offering to globalize their
processes and make it easier to build and track submissions content
through approvals. Submission preparations are fast and efficient, while
meeting global compliance requirements.
Improved audit readiness in quality
ICON is leveraging Vault QualityDocs to enable efficient routine audits
for its quality assurance department. The solution provides real-time
access to information, reducing compliance risk and improving quality
processes. In addition, ICON is now able to expand its service
offerings, including support for remote audits – a service clients are
requesting more often.
“Veeva Vault is a game-changer for many companies looking to unify
processes in regulatory, quality, clinical, and beyond,” said Jennifer
Goldsmith, senior vice president of Veeva Vault. “As the level of
activity between sponsors and contract services organizations continues
to increase, customers such as ICON will be able to provide greater
visibility, efficiency, and compliance for their sponsors.”
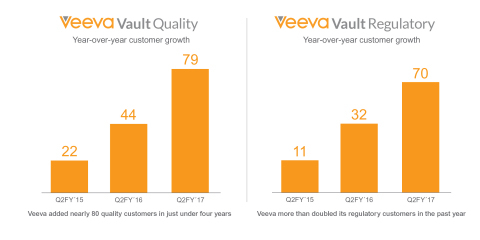
In other news today, Veeva announced broad industry adoption of its Veeva
Vault Quality applications. Read our press
release to learn more.
Additional Information
For more on Veeva Vault Submissions,
visit: veeva.com/Submissions
For
more on Veeva Vault QualityDocs, visit: veeva.com/QualityDocs
Connect
with Veeva on LinkedIn: linkedin.com/company/veeva-systems
Follow
us @veevasystems on Twitter: twitter.com/veevasystems
Like
Veeva on Facebook: facebook.com/veevasystems
About Veeva Systems
Veeva Systems Inc. is a leader in
cloud-based software for the global life sciences industry. Committed to
innovation, product excellence, and customer success, Veeva has more
than 450 customers, ranging from the world's largest pharmaceutical
companies to emerging biotechs. Veeva is headquartered in the San
Francisco Bay Area, with offices in Europe, Asia, and Latin America. For
more information, visit www.veeva.com.
Forward-looking Statements
This release contains
forward-looking statements, including the market demand for and
acceptance of Veeva’s products and services, the results from use of
Veeva’s products and services, and general business conditions,
particularly in the life sciences industry. Any forward-looking
statements contained in this press release are based upon Veeva’s
historical performance and its current plans, estimates, and
expectations, and are not a representation that such plans, estimates,
or expectations will be achieved. These forward-looking statements
represent Veeva’s expectations as of the date of this press
announcement. Subsequent events may cause these expectations to change,
and Veeva disclaims any obligation to update the forward-looking
statements in the future. These forward-looking statements are subject
to known and unknown risks and uncertainties that may cause actual
results to differ materially. Additional risks and uncertainties that
could affect Veeva’s financial results are included under the captions,
“Risk Factors” and “Management’s Discussion and Analysis of Financial
Condition and Results of Operations,” in the company’s filing on Form
10-Q for the period ended July 31, 2016. This is available on the
company’s website at veeva.com
under the Investors section and on the SEC’s website at sec.gov.
Further information on potential risks that could affect actual results
will be included in other filings Veeva makes with the SEC from time to
time.

View source version on businesswire.com: http://www.businesswire.com/news/home/20161018005699/en/
Source: Veeva Systems Inc.